Introduction
Garlic (Allium sativum L.), a herbaceous plant belonging to the Liliaceae family, has been widely cultivated and utilized worldwide for its dual roles as a spice and medicinal ingredient. Its health-promoting properties are largely attributed to the rich bioactive compounds it contains, such as sulfur-containing compounds, flavonoids, and saponins, which contribute to its diverse physiological activities (Kim et al., 2009; Sunanta et al., 2023). These bioactive compounds have been shown to induce potent antioxidant effects, which help reduce oxidative stress, a key factor in the pathogenesis of chronic diseases (Kim et al., 2010; El-Saadony et al., 2024). Additionally, garlic exhibits significant antimicrobial activities, effectively inhibiting the growth of various pathogens (Mozaffari et al., 2014). Its anticancer potential has been highlighted through studies demonstrating its ability to suppress tumor growth and induce apoptosis in cancer cells (Banerjee & Maulik, 2002; Sunanta et al., 2023). Furthermore, garlic is recognized for its cardiovascular benefits, including anticoagulant and cholesterol-lowering effects, which contribute to improved heart health (Masjedi et al., 2013; Qidwa & Ashfaq, 2013). These properties underscore the potential of garlic as a functional food ingredient with wide-ranging health benefits (Kyriacou et al., 2016). Despite its many advantages, garlic experiences significant difficulties during post-harvest storage. Prolonged storage often leads to quality decline, such as sprouting or decay, which not only reduces its market value but also generates economic losses (Logan et al., 1996). Sprouting, while considered a sign of spoilage in conventional uses, may enhance the bioactivity of garlic by increasing its levels of certain bioactive compounds, such as S-allyl cysteine and flavonoids (Shang et al., 2019). These findings suggest that sprouted garlic, rather than being discarded, could be re-evaluated for its potential as a high-value functional ingredient.
In South Korea, the garlic industry faces periodic supply-demand imbalances, compounded by competition from low-cost imports, particularly from China. These supply-demand challenges have prompted efforts to develop alternative uses and value-added applications for garlic beyond its traditional uses (Choi et al., 2005; Lee et al., 2007). One such strategy includes green garlic, harvested for its tender leaves during the early stages of growth, which is noted for its nutritional and functional properties (Kim & Chung, 1997). Another approach involves the cultivation of light-deprived garlic under dark conditions to produce leaves with unique biochemical compositions (Choi et al., 2005). However, while these alternative applications highlight the multifunctionality of garlic, the potential of sprouted garlic as a functional food ingredient remains understudied (Logan et al., 1996; Ruddock et al., 2005).
A growing consumer preference for functional foods is being driven by an increased focus on health, well-being, and sustainable lifestyles, as indicated by recent trends (Ha et al., 2009; Kyriacou et al., 2016; Moses et al., 2024). Sprouted vegetables, in particular, have gained attention due to their enriched nutritional profiles, which show higher concentrations of amino acids, vitamins, minerals, enzymes, and bioactive compounds (Khalil et al., 2007; Melguizo-Rodríguez et al., 2022). Studies on the sprouts of oats, barley, and broccoli have demonstrated their potential health benefits, such as improved antioxidant capacity and enhanced bioavailability of nutrients (Kyriacou et al., 2016). However, there is limited research on sprouts of garlic (Sunanta et al., 2023). Due to the increasing demand for innovative functional food products, there is a pressing need to investigate the biochemical composition and health benefits of sprouted garlic. We hypothesize that hydroponically cultivated sprouted garlic undergoes biochemical changes that enhance its functional food properties, particularly in terms of antioxidant capacity and nutrient composition. This study aims to analyze the biochemical and bioactive properties of sprouted garlic cultivated under hydroponic conditions to evaluate their potential as functional foods. These findings could provide both nutritional and economic benefits, offering a sustainable solution to post-harvest problems while meeting the demands of the functional food market.
Materials and Methods
The garlic was harvested in 2023 from Uiseong (Gyeongsangnam-do, Korea), and purchased from the Hanaro Club in Goyang (Gyeonggi-do, Korea).
Garlic cloves weighing 6 g each were cultivated hydroponically under non-circulating conditions at 17°C and 45% relative humidity for 20 days. Growth measurements were taken every 4 days (days 0, 4, 8, 12, 16, and 20). The harvested samples were immediately freeze-dried using a Coolsafe 95-15 Pro freeze dryer (Scanvac, Lillerød, Denmark) for further analysis.
Growth measurements were performed every 4 days during the hydroponic cultivation period. The lengths of sprouts and roots from ten garlic cloves were measured using a Vernier caliper (CD-P30S, Mitutoyo, Kanagawa, Japan). The weights of the garlic samples were determined using a balance (OHAUS PAG214C, Parsippany, NJ, USA) after carefully removing surface water.
The proximate composition of freeze-dried and powdered samples was analyzed according to AOAC methods (2001). Moisture content was determined by oven drying using an OF-22GW drying oven (Jeio Tech Co., Ltd., Daejeon, Korea) at 105°C until a constant weight was obtained. Crude protein content was analyzed through the Micro-Kjeldahl method. Crude fat content was analyzed through Soxhlet extraction. Ash content was determined by dry ashing in an electric muffle furnace (EYELA, TMF-2100, Tokyo, Japan) at 550°C. Carbohydrate content was calculated by subtracting the total percentages of moisture, protein, fat, and ash from 100%.
Mineral content analysis was conducted by decomposing a 0.1 g freeze-dried and powdered sample with 10 mL of 50% nitric acid. The samples were digested using a microwave digestion system (MARS 5, CEM Corp., Matthews, NC, USA), filtered, and diluted to a final volume of 100 mL with deionized water. The mineral contents were analyzed using inductively coupled plasma optical emission spectroscopy (ICP-OES; Optima 7300 V, Perkin Elmer Instruments Co., Ltd., Waltham, USA; NFEC-2023-07-289084) at the Biopolymer Research Center for Advanced Material (BRCAM, Sejong University, Seoul, Korea).
One gram of freeze-dried and powdered sample was combined with 100 times its weight in deionized water. The mixture was sonicated for 30 min to extract free amino acids. The extract was filtered through Whatman No. 1 filter paper and centrifuged at 10,000×g for 10 min. The upper layer was filtered through a 0.45 μm membrane filter for high-performance liquid chromatography (HPLC) analysis. Pre-column derivatization was performed using derivatization reagents (Agilent 5061-3339, 5061-3335, 5061-3337; Agilent Technologies, Santa Clara, CA, USA) following the method by Chirinang & Intarapichet (2009). The derivatization mixture was prepared with 5.0 μL of 0.1 M borate buffer, 1.0 μL of the sample, 1.0 μL of 3-mercaptopropionic acid (3-MPA)/o-phthalaldehyde (OPA) reagent, and 1.0 μL of 9-fluorenylmethyl chloroformate (FMOC-Cl) reagent, followed by dilution with 32 μL of water. Amino acid analysis was performed using a Dionex Ultimate 3000 HPLC system (Thermo Dionex, Pleasanton, CA, USA) equipped with an Agilent 1260 Infinity FL detector (Agilent, Palo Alto, CA, USA) at a detection wavelength of 340 nm. An Inno C18 column (4.6×150 mm, 5 μm; Youngjin Biochrom, Seongnam, Korea) was used, and the mobile phases consisted of 40 mM sodium phosphate buffer (pH 7.0) as eluent A, and a mixture of water:acetonitrile:methanol (10:45:45, v/v/v%) as eluent B. The flow rate was 1.5 mL/min. The initial condition was 95% eluent A for 3 min, followed by a linear gradient to 55% eluent B within 1 min, then 90% eluent B for 6 min. Washing and equilibration were performed at 5% eluent B. The column temperature was maintained at 40°C. The concentration of each amino acid was calculated based on a standard curve prepared using an amino acid standard mixture (Agilent 5061-3330; Agilent Technologies, Santa Clara, CA, USA).
The enzyme activities were determined using extracts prepared by adding deionized water at 100 times the weight of freeze-dried and powdered samples, followed by sonication for 30 min. The extract was filtered through Whatman No. 1 filter paper and centrifuged at 10,000×g for 10 min. The supernatant was used for analysis. Protease activity was measured using azo-casein (S-AZCAS 12/07, Megazyme, Bray, Ireland) as a substrate (Ndidi & Nzelibe, 2012). The reaction was observed by measuring absorbance at 440 nm. The protease activity was calculated using the equation:
(R=0.99, Papain from Papaya latex, pH 7.0).
Amylase activity was evaluated using 0.5% soluble starch as a substrate. The glucose produced was quantified using a YSI biochemistry analyzer (YSI 2700 SELECT™, YSI Inc., Yellow Springs, OH, USA). This method enabled the determination of amylase activity based on the enzymatic hydrolysis of starch and subsequent glucose production.
Ten grams of freeze-dried and powdered garlic was extracted twice using 50 mL of 80% ethanol at 80°C for 1 h. The ethanol was removed, and the extract was concentrated using a rotary evaporator (R-124, Büchi Labortechnik, Flawil, Switzerland). The concentrated extract was diluted to a final volume of 10 mL with deionized water and filtered through a 0.45 μm membrane filter. Vitamin C was quantified using a Dionex Ultimate 3000 HPLC System (Thermo Fisher Scientific, Pleasanton, CA, USA) equipped with a UV/VIS detector (λ=254 nm; Spectra Physics, Milpitas, CA, USA). A μ-Bondapak C18 column (3.9×300 mm, 10 μm; Waters Assoc., Milford, MA, USA) was utilized. The mobile phase consisted of 50 mM KH2PO4:acetonitrile (60:40, v/v%) with a flow rate of 1.0 mL/min. The column temperature was maintained at 30°C. Ascorbic acid (Junsei Chemical Co., Ltd, Tokyo, Japan) was used as the standard.
The total polyphenol content was determined using the Folin-Ciocalteu method (Zakarova et al, 2014). One gram of freeze- dried and powdered sample was extracted with deionized water (100 times the sample weight) via sonication for 30 min. The extract was filtered through Whatman No. 1 filter paper and centrifuged at 10,000×g for 10 min. The supernatant was used for analysis. Total polyphenol content was expressed as gallic acid equivalents (GAE) based on a standard curve prepared using gallic acid standards.
The radical scavenging abilities of garlic sprout extracts were assessed using the DPPH and ABTS assays. Freeze-dried and powdered sprouted garlic were extracted using the previously described sonication method. After filtration and centrifugation, the supernatant was analyzed as follows:
DPPH radical scavenging activity was measured using a modified Blois method (1958). The absorbance of the reaction mixture was measured at 517 nm, and the percentage of scavenging activity was calculated by comparing the absorbance of the sample to that of the control.
ABTS radical scavenging activity was determined using a modified ABTS cation decolorization assay (Jang et al., 2018). The absorbance of the reaction mixture was measured at 734 nm, and the percentage of scavenging activity was calculated by comparing the absorbance of the sample to that of the control.
All experiments were performed in triplicate, and the results are presented as mean±standard deviation. Statistical analysis was conducted using one-way analysis of variance (ANOVA) with the SPSS software (Version 23.0, SPSS Inc., Chicago, IL, USA). Significant differences among mean values were determined using Duncan’s multiple range test at a significance level of p<0.05.
Results and Discussion
The growth characteristics of garlic during hydroponic cultivation are summarized in Table 1. A significant increase in both sprout length and weight was observed as the cultivation period progressed. The sprout length reached 14.14±5.19 mm, and the weight increased to 6.69±0.83 g by day 8, representing a 1.2-fold increase in weight compared to day 4. By day 20, the sprout length and weight had increased approximately 6.9-fold (97.94±22.47 mm) and 1.4-fold (9.11±1.32 g) compared to day 4, highlighting the rapid growth during the early and intermediate stages of hydroponic cultivation. The rapid growth observed in sprouted garlic during the early stages of hydroponic cultivation (days 4–8) aligns with findings in sprouted barley and broccoli, where nutrient mobilization and enzymatic activity significantly influence initial shoot elongation (Kim et al., 2013; Choi et al., 2015). However, unlike barley sprouts, which exhibit more rapid biomass accumulation due to their high carbohydrate reserves, sprouted garlic shows a slower but steady increase in weight, possibly due to its reliance on sulfur-containing metabolites (Kyriacou et al., 2016). This suggests that metabolic adaptations in garlic may favor bioactive compound accumulation over rapid structural growth. The early growth acceleration observed between days 4 and 8 is consistent with previous studies on other sprouted vegetables such as barley and radish, which also demonstrated rapid nutrient mobilization during early growth stages (Kim et al., 2013; Choi et al., 2015). This suggests that garlic, like other sprouted vegetables, changes stored nutrients within the cloves to support initial sprout development. The increase in sprout length during this period may reflect the efficient conversion of stored carbohydrates into energy, driving cellular expansion and division (Park et al., 2015). The growth rate plateaued, with sprout weight showing only a marginal increase at 20 days. This stabilization may indicate the depletion of readily available stored nutrients in the garlic cloves, as also observed in peanut and broccoli sprouts during later stages of hydroponic cultivation (Xu et al., 2005; Lee et al., 2007). The reduced rate of weight gain, despite the continued elongation of the sprout, suggests a shift in metabolic focus from biomass accumulation to structural development, possibly involving the synthesis of secondary metabolites that contribute to the functional properties of the sprouts (Kyriacou et al., 2016). These results demonstrate the dynamic growth pattern of sprouted garlic under hydroponic conditions, emphasizing the importance of the early cultivation period (days 4–8) for maximizing biomass production. The significant growth observed during this period suggests that optimizing nutrient supply and environmental factors during early stages could enhance sprout quality and yield. Additionally, the comparable growth trends between garlic and other sprouted vegetables highlight the broader applicability of hydroponic cultivation techniques in functional food production.
The proximate composition of sprouted garlic was changed throughout the hydroponic cultivation period (Table 2). The moisture content showed an initial decrease from 7.38±0.07% on day 4 to 3.20±0.09%. However, by day 20, the moisture content increased slightly to 5.13±0.12%. Protein content increased significantly from 17.73% to 20.21%, indicating enhanced protein synthesis during sprouting. This increase is likely associated with the production of enzymes and structural proteins necessary for metabolic processes and sprout development. It has been reported in other sprouted vegetables, such as mung beans and barley, where enhanced enzymatic activity contributes to nutrient mobilization and metabolic regulation (Saikusa et al., 1994). The observed increase in protein content (17.73% to 20.21%) during sprouting is consistent with findings in sprouted barley and broccoli, where protein biosynthesis is upregulated to support enzymatic functions during germination (Saikusa et al., 1994). In contrast, sprouted barley exhibits a significant increase in total protein due to enhanced storage protein mobilization, whereas sprouted garlic appears to rely more on metabolic enzyme synthesis, reflecting a different nutrient allocation strategy. Similarly, the gradual increase in fat content in sprouted garlic is in line with sprouted broccoli, where lipid metabolism shifts towards polyunsaturated fatty acid synthesis, enhancing the functional properties of the sprouts (Melguizo-Rodríguez et al., 2022). The fat content, which remained relatively stable during the early cultivation period, showed a significant increase from 0.43±0.02% on day 4 to 0.76±0.02% on day 20. This suggests the accumulation of lipids during later stages, potentially serving as an energy reservoir for sustained metabolic activity. Interestingly, the slight reduction in fat content observed before day 4 could indicate its utilization as an energy source to support the initiation of sprouting. The ash content showed variation throughout the cultivation period, with an initial increase followed by a slight decline by day 20. These variations may reflect the alterations in the redistribution of inorganic nutrients essential for cellular and metabolic functions during sprout development. In contrast, the carbohydrate content displayed an inverse pattern, with contents increasing initially but subsequently decreasing during sprout growth. This decline matches the utilization of stored carbohydrates to support rapid growth and metabolic processes, consistent with findings in other sprouted crops (Kim et al., 2013).
When compared to the standard composition of garlic, 7.3% moisture, 15.3% protein, 0.5% fat, and 73.7% carbohydrates (Rural Development Administration. 2016), the observed variations in nutrient content may be attributed to differences in production years, and cultivation conditions. The proximate composition analysis demonstrates changes in nutrient distribution during the sprouting process, highlighting the need to optimize cultivation conditions to enhance the nutritional quality of sprouted garlic. The higher protein and fat contents in sprouted garlic highlight the metabolic change during germination. These results are consistent with known metabolic patterns in sprouted crops, further validating the potential of sprouted garlic via hydroponic cultivation as a nutrient-rich functional food.
The mineral composition of sprouted garlic showed changes throughout the hydroponic cultivation period, reflecting the utilization of nutrients essential for sprout development (Table 3). The phosphorus, sodium, and potassium contents steadily increased until day 16, underscoring their pivotal roles in cellular processes such as energy transfer and osmoregulation, which are critical for plant growth (Prajapati & Modi, 2012). In contrast, magnesium and iron reached their maximum, earlier, with magnesium on day 12 (786.05±3.52 mg/kg) and iron on day 8 (49.07±1.05 mg/kg), followed by declines as sprout development progressed. The magnesium and iron content suggest their importance during the initial stages of sprouting. Magnesium plays a vital role in chlorophyll synthesis and enzymatic activation, which are critical for early metabolic activities (Terry & Low, 1982). Similarly, iron is directly related to chlorophyll content and thylakoid synthesis, further emphasizing its necessity during the early growth phase. The decline in magnesium and iron content likely indicates their consumption as metabolic activities stabilize in the later stages of sprout growth. Sodium and phosphorus contents showed notable increases until day 8 and day 12, respectively, before declining. Sodium contributes to maintaining cellular turgor and nutrient transport, while phosphorus is indispensable for energy metabolism and the synthesis of nucleic acids. These results suggest that both elements are actively utilized during the rapid growth phases of garlic sprouts. Potassium content exhibited a consistent increase until day 12, stabilizing thereafter, which may indicate its essential but limited role in the sprout structure is established. Potassium was involved in stomatal regulation and enzyme activation in the early stage of development but suggests that additional uptake is unnecessary after a certain stage of growth. The fluctuation in zinc content, characterized by repeated increases and decreases, indicates its highly responsive role during garlic sprout development. Zinc functions as a cofactor for various enzymes and contributes to protein synthesis, which is critical for cellular proliferation and structural development during sprouting. These variations in zinc content suggest a change in demand depending on specific growth phases.
These mineral concentrations are consistent with findings in other sprouted vegetables, such as barley and radish, where nutrient consumption and redistribution support rapid growth and structural formation (Gibson et al., 2001). The use of tap water in this study may have influenced the availability of certain minerals, potentially limiting the full growth potential of sprouted garlic. Future studies using nutrient-enriched solutions could provide deeper insights into optimizing mineral uptake and improving the nutritional quality of sprouted garlic through hydroponic cultivation. Therefore, the mineral composition analysis highlights the importance of precise nutrient management during hydroponic cultivation to ensure optimal growth and functional quality of sprouted garlic. These findings not only enhance our understanding of nutrient dynamics during sprouting but also underscore the potential of sprouted garlic as a nutrient-dense functional food.
The free amino acid composition of sprouted garlic exhibited significant changes throughout the hydroponic cultivation period, as shown in Table 4. The total content of free amino acids, including both non-essential and essential amino acids, displayed a consistent upward trend, increasing from 5,301.21 mg% at the initiation of cultivation to 6,962.80 mg% after 20 days. This substantial increase highlights the dynamic biochemical transformations occurring during sprouting, driven by enhanced enzymatic activity and protein metabolism. Among the individual amino acids, arginine (2,417.72 mg%) and glutamine (2,261.52 mg%) were the most abundant in sprouted garlic after 20 days, collectively accounting for 67.2% of the total amino acid content. Arginine is known for its role in nitrogen metabolism and as a precursor for the synthesis of polyamines, which are critical for cellular growth and stress responses (Saikusa et al., 1994). Glutamine, on the other hand, serves as a key substrate for energy production and plays an essential role in flavor development through its interaction with enzymes such as glutaminase. The marked increase in glutamine content suggests its potential for enhancing the sensory qualities of food products through enzymatic processing.
The elevation in total free amino acids corresponds to the increase in protein content observed in the proximate composition analysis, reinforcing the role of proteolytic enzymes in breaking down storage proteins into free amino acids during sprouting. Similar trends have been reported in other sprouted vegetables, such as mung beans and pumpkin seeds, where proteolytic activity drives the accumulation of free amino acids during germination (Lee et al., 2003). This process is integral to the nutritional and functional properties of sprouts, as free amino acids are highly bioavailable and contribute significantly to the nutritional value of food products (Cho & Yang, 2007). Essential amino acids, including lysine, threonine, and valine, also showed substantial increases during the cultivation period, highlighting the enhanced nutritional quality of hydroponically grown garlic. These amino acids are crucial for human health, particularly for protein synthesis and metabolic regulation. The increased levels of both essential and non-essential amino acids underscore the potential of sprouted garlic as a functional food ingredient with enhanced nutritional benefits.
The observed trends in free amino acid composition not only reflect the metabolic shifts associated with sprouting but also have significant implications for the sensory and nutritional quality of sprouted garlic. The elevated glutamine and arginine levels suggest that hydroponically cultivated garlic could serve as a valuable ingredient for flavor enhancement and functional food applications. Moreover, the similarity of these findings to observations in other sprouted vegetables supports the application of hydroponic cultivation for enhancing the nutritional composition of sprouted crops.
The activities of protease and amylase, key enzymes involved in nutrient mobilization, fluctuated significantly throughout the hydroponic cultivation period of sprouted garlic (Table 5). These fluctuations reflect the dynamic metabolic adjustments required to support sprout growth and development.
Protease activity increased markedly from 12.33±1.29 mU/mL on day 0 to 20.79±0.59 mU/mL by day 4, indicating enhanced protein degradation during the early sprouting phase. This early increase likely reflects the breakdown of storage proteins into free amino acids, which serve as building blocks for new protein synthesis and energy metabolism during rapid growth (days 0–8). However, protease activity decreased sharply to 5.40±0.00 mU/mL on day 12, possibly due to reduced demand for protein degradation as the sprout structure becomes more established. Interestingly, protease activity surged again to 21.26±2.47 mU/mL on day 20, suggesting a secondary phase of protein metabolism, potentially associated with the synthesis of enzymes or structural proteins needed for late-stage sprout maturation.
Amylase activity, responsible for the hydrolysis of starch into simple sugars, showed a different pattern. After an initial decline from 194.82±4.73 mU/mL on day 0 to 170.00±1.67 mU/mL by day 4, amylase activity steadily increased, peaking at 299.82±0.85 mU/mL on day 20. This trend suggests that starch breakdown becomes more critical as the sprouts mature, providing a continuous supply of glucose to meet the increasing energy demands during later stages of growth. The initial decrease may indicate that stored carbohydrates are sufficient to meet the metabolic needs during the very early sprouting phase, with amylase activity ramping up only as those reserves are depleted.
Variations in protease and amylase activities reflect metabolic patterns reported in other sprouted crops, such as barley and mung beans, where enzymatic activity adjusts dynamically to meet the developmental and metabolic demands of sprouting (Saikusa et al., 1994; Hwang et al., 2019). The biphasic pattern of protease activity suggests a tightly regulated process of protein metabolism, tailored to different stages of sprout development. Similarly, the gradual increase in amylase activity highlights the central role of carbohydrate mobilization in sustaining growth during the later phases of sprouting. The interplay between protease and amylase activities underscores the importance of coordinated nutrient mobilization during sprout development. Early protease activity supports rapid cellular growth by providing essential amino acids, while increased amylase activity in later stages ensures a steady energy supply through starch hydrolysis. These findings emphasize the critical role of enzymatic regulation in optimizing the nutritional and functional quality of sprouted garlic. Future research could explore the impact of environmental factors such as temperature and nutrient availability on enzymatic activity to further optimize hydroponic cultivation conditions. Understanding these dynamics could enhance the production of sprouted garlic with higher nutritional and functional properties, catering to the growing demand for high-value functional foods.
Vitamin C is a well-known antioxidant that plays a critical role in neutralizing reactive oxygen species (ROS) and inhibiting lipid peroxidation in biological systems (Davey et al., 2007; Lee et al., 2012). The changes in vitamin C content during the hydroponic cultivation of garlic are presented in Fig. 1. The vitamin C content increased significantly from day 0 (196.23 mg/kg) to day 8, reaching a peak of 307.25 mg/kg (day 4: 256.28 mg/kg; day 8: 307.25 mg/kg). The most substantial increase occurred during the first 4 days of cultivation, with an approximate 30% increment, followed by an additional 20% increase between days 4 and 8. This result suggests that vitamin C synthesis is most active during the initial sprouting phase, supporting the early development of garlic sprouts. However, after day 8, vitamin C levels began to decline, decreasing to 238.70 mg/kg on day 12, 215.36 mg/kg on day 16, and ultimately 191.24 mg/kg by day 20. This decline may indicate that the rate of vitamin C utilization for metabolic processes and growth exceeded its rate of synthesis during the later stages of cultivation. Such a pattern is consistent with previous findings in other sprouted crops, where vitamin C levels peak during early growth and subsequently decrease as the sprouts mature (Logan et al., 1996; Xu et al., 2005).
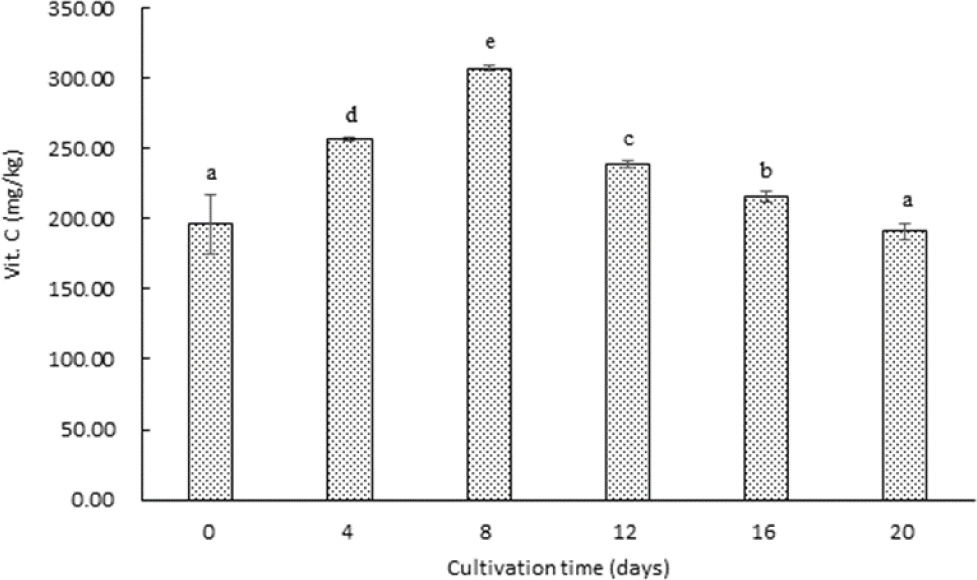
The elevated vitamin C content observed in hydroponically cultivated garlic during the early growth phase highlights its potential as a functional food with enhanced nutritional value. Notably, the peak vitamin C content (307.25 mg/kg) surpasses that of regular garlic, emphasizing the benefits of hydroponic germination for boosting antioxidant levels. This enhanced antioxidant capacity may contribute to the overall health-promoting properties of sprouted garlic, particularly in combating oxidative stress-related conditions. The synthesis and accumulation of vitamin C in sprouts are influenced by environmental factors such as light quality, intensity, and duration. Previous studies have demonstrated that illumination type, including light color and irradiance, can significantly affect vitamin C content during sprout cultivation (Foyer, 1991; Logan et al., 1996; Kim et al., 2012; Choi et al., 2015). Given the decline in vitamin C content during the later stages of garlic sprout growth, optimizing light conditions during hydroponic cultivation could enhance vitamin C retention and overall sprout quality. Further research is warranted to investigate the effects of various lighting conditions on vitamin C synthesis and accumulation in sprouted garlic. Specifically, exploring the impact of different light spectra and intensities could provide valuable insights into maximizing the nutritional quality of hydroponically cultivated garlic. Additionally, studies on the interaction between light and other environmental factors, such as temperature and medium composition, could help refine cultivation techniques for functional food production.
Phenolic compounds, recognized for their potent antioxidant properties, play a vital role as scavengers of free radicals due to their free phenolic hydroxyl groups (Ruddock et al., 2005). These compounds are abundant in various plant-based foods and are particularly enriched in sprouted crops, which are associated with numerous health benefits (Kyriacou et al., 2016). In this study, the phenolic compound content in garlic sprouted garlic through hydroponic cultivation was analyzed, and the results are presented in Fig. 2. The phenolic content of sprouted garlic demonstrated an increase during the 12 days of hydroponic cultivation, maximum at 42.15±0.87 mg/100 g. This early-stage increase suggests enhanced biosynthesis and accumulation of phenolic compounds during the rapid growth phase of sprouted garlic. The phenolic content showed a slight decline at 38.62±0.64 mg/100 g from 12 day to 20 day. This pattern indicates that phenolic compounds are actively synthesized during the early stages of sprouting but may be partially degraded or utilized in metabolic processes as the sprouts mature. The accumulation of phenolic compounds in sprouted garlic reached a peak at day 12 (42.15 mg/100 g), a trend also observed in sprouted broccoli, where phenolic content increases during early germination and stabilizes at later stages (Kyriacou et al., 2016). However, while broccoli sprouts exhibit a higher total phenolic content due to their cruciferous origin, sprouted garlic contains unique sulfur-containing phenolic derivatives that contribute to its distinct antioxidant properties. This differentiation suggests that the bioactive potential of sprouted garlic is not solely dependent on phenolic content but also on the synergistic effects of sulfur metabolites and flavonoids.
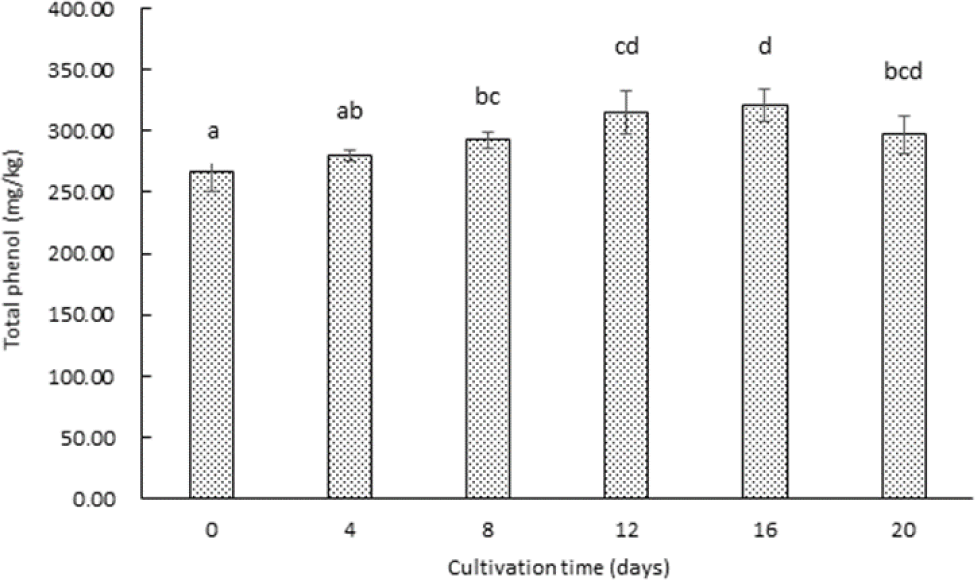
Previous studies analyzing garlic cultivated in major garlic-producing regions of South Korea, including Taean, Uiseong, Seosan, Jeju, Namhae, Hampyeong, Muan, and Hapcheon, reported that the total phenolic contents ranged from 12.69±0.18 to 22.02±0.27 mg/100 g. The phenolic content of garlic used in this study was slightly higher, likely due to differences in cultivation environments, such as soil composition, climate, and cultivation method. This variability highlights the influence of environmental factors on the phenolic profile of garlic and underscores the potential for hydroponic cultivation to enhance phenolic content through controlled growth conditions. The enrichment of phenolic compounds in sprouted garlic compared to non-sprouted garlic underscores their potential as a high-value functional food. Phenolic compounds contribute to the health-promoting properties of sprouted garlic, particularly through their antioxidant capacity, which can mitigate oxidative stress and reduce the risk of chronic diseases. The accumulation of phenolic compounds during the early stages of hydroponic cultivation suggests that optimizing growth conditions during this period could maximize their functional properties. Variations in phenolic content based on different extraction solvents have been reported, emphasizing the need for standardized extraction protocols to accurately evaluate the phenolic profile of sprouted garlic (Chae et al., 2019). Future studies could determine the influence of various extraction solvents and methodologies on phenolic compound yield to provide a more comprehensive understanding of their bioavailability and functional properties. Additionally, investigating the impact of other environmental factors, such as light quality and medium composition, on phenolic biosynthesis during hydroponic cultivation could further enhance the antioxidant potential of sprouted garlic.
Antioxidant activity is a crucial indicator of the health-promoting potential of plant-based foods, with assays such as DPPH and ABTS commonly used to evaluate free radical scavenging ability. These assays provide the capacity of antioxidants to neutralize reactive oxygen species and mitigate oxidative stress (Hwang et al., 2019). In this study, the antioxidant activity of sprouted garlic via hydroponic cultivation was analyzed using DPPH and ABTS assays, as shown in Fig. 3A, B. The DPPH radical scavenging activity of sprouted garlic showed minimal variation during the 8 days of hydroponic cultivation. However, a noticeable increase was observed with the sprout emergence and active growth phases. The DPPH radical scavenging ability reached its maximum by day 20, reflecting the enhanced antioxidant capacity of sprouted garlic during the later stages of cultivation. This result is similar to previous studies, where the radical scavenging ability was strongly correlated with the accumulation of phenolic compounds in the natural products (Choi et al., 2007). The increase in phenolic compounds observed in sprouted garlic likely contributed to its enhanced DPPH radical scavenging activity, underscoring the role of these compounds as key contributors to antioxidant capacity. The ABTS radical scavenging activity of sprouted garlic followed a pattern similar to the changes in their phenolic compound content (Fig. 3B). The activity increased significantly until day 12, reaching its maximum during the early growth phase. The phenolic compounds are well-known for their electron-donating properties, which neutralize ABTS radicals. Therefore, this increase suggested a strong correlation between phenolic content and ABTS radical scavenging ability. A gradual decline in ABTS activity was observed after day 12, although the changes were not statistically significant. This decline may reflect a stabilization of antioxidant compound synthesis as the sprouts matured, with phenolic compounds being utilized in metabolic processes or undergoing degradation.
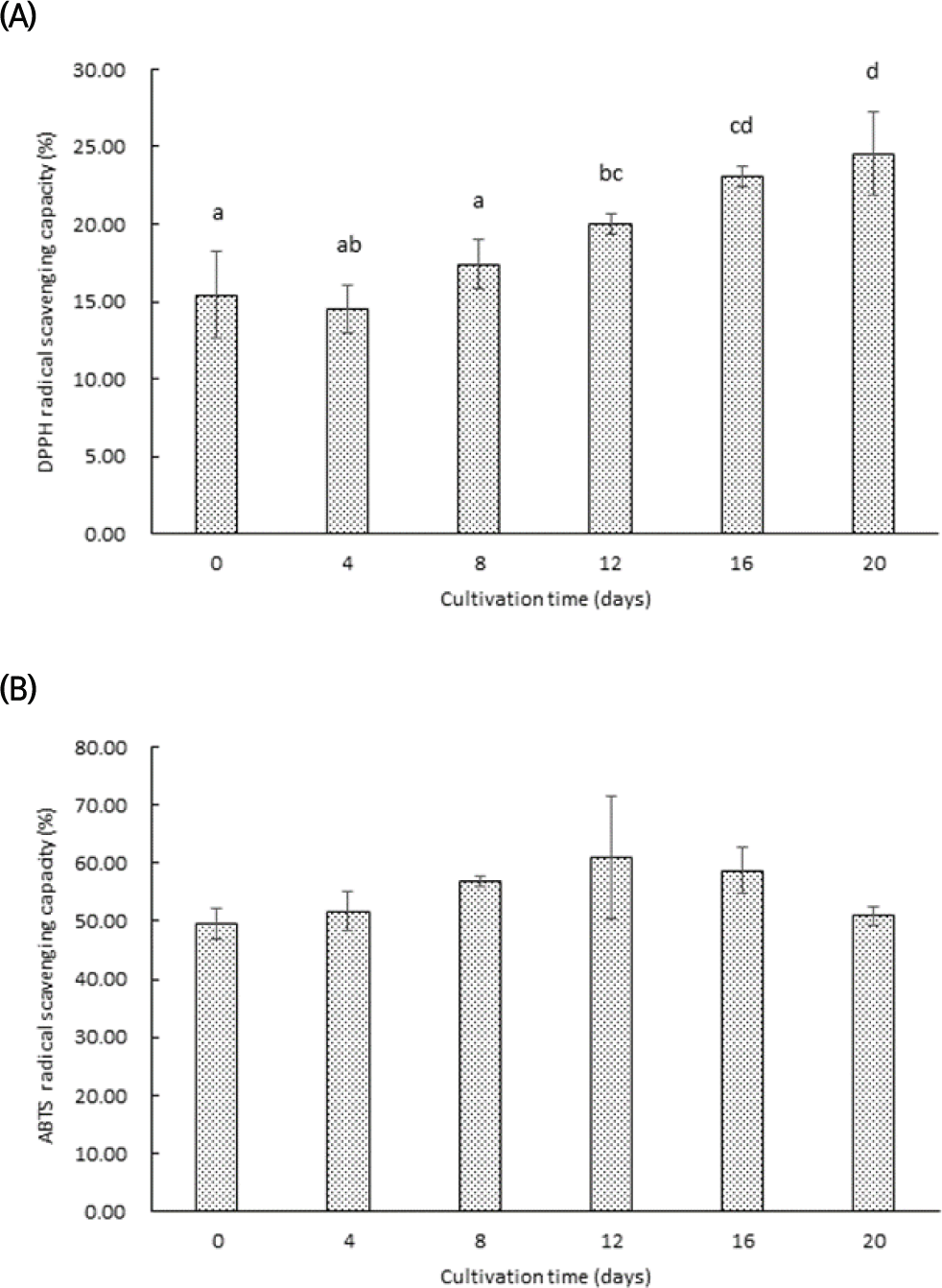
The antioxidant activity of sprouted garlic, as determined by DPPH and ABTS assays, followed a similar pattern to that observed in sprouted broccoli, where the highest radical scavenging capacity coincided with the maximum phenolic content (Jang et al., 2018). However, the radical scavenging ability of sprouted garlic appeared to be more sustained over time, suggesting that sulfur-containing compounds, in addition to polyphenols, play a significant role in its antioxidant potential. Compared to sprouted barley, which primarily exhibits antioxidant activity through flavonoids, sprouted garlic benefits from a broader spectrum of bioactive compounds, including organosulfur compounds, further enhancing its functional food potential. The similar results were observed in both DPPH and ABTS assays. It was a strong association between phenolic compound content and antioxidant activity in sprouted garlic. The enhanced antioxidant capacity during the early and mid-stages of hydroponic cultivation suggests that optimizing growth conditions during these periods could maximize the health-promoting properties of sprouted garlic. Additionally, the DPPH and ABTS activities observed in the later stages indicate the potential for sprouted garlic to retain substantial antioxidant activity even as they mature. To further enhance the antioxidant potential of sprouted garlic, future studies should investigate the impact of environmental factors such as light intensity, medium composition, and hydroponic system modifications on antioxidant compound synthesis.
Conclusion
This study presents an analysis of the nutrient composition and functional properties of sprouted garlic cultivated hydroponically, providing valuable insights into its potential as a functional food. Morphological changes were observed, with root initiation occurring by day 4 and an increase in sprout length and weight by day 8. Proximate composition analysis demonstrated changes in the contents of protein, fat, ash, and carbohydrates. Specifically, fat was utilized during sprout initiation, while carbohydrates supported subsequent growth, emphasizing the metabolic adaptability of garlic during germination. Mineral content analysis indicated variations in the concentrations of phosphorus, sodium, potassium, magnesium, iron, and zinc. These patterns suggested nutrient uptake during the early sprouting phase and their later use to support metabolic and structural development. Furthermore, free amino acids, especially arginine and glutamine, were increased, thereby enhancing the nutrition and flavor profile of sprouted garlic. Vitamin C content increased during the early sprouting phase, reflecting active synthesis, followed by a decline as it was utilized for growth processes. This pattern underscores the importance of the early cultivation phase in maximizing antioxidant properties. Similarly, phenolic compounds, known for their significant contribution to antioxidant activity, were enriched during the 12 days of cultivation, confirming sprouted garlic as a potential source of natural antioxidants. Antioxidant activities, evaluated through DPPH and ABTS assays, supported these findings, further reinforcing the potential of sprouted garlic as a functional food with enhanced health-promoting properties.
To maximize the nutritional and functional potential of sprouted garlic, future research should focus on optimizing hydroponic cultivation conditions, including formulation of medium, light quality, and growth duration, to enhance the accumulation of bioactive compounds and antioxidant capacity. Moreover, examining the impact of diverse environmental and cultivation parameters on flavor profiles and nutrient preservation would provide critical insights for the development of commercially viable cultivation strategies. In conclusion, sprouted garlic cultivated hydroponically demonstrates substantial nutritional and functional potential, making it a viable candidate for inclusion in the growing market for health-oriented functional foods. Its enriched bioactive compounds, coupled with its flavor and antioxidant properties, establish it as a valuable addition to both the food and nutraceutical industries.
